Industry
Services
Highlights
- The pharmaceutical industry is revolutionising regulatory affairs by adopting advanced technologies like agentic AI, a shift encouraged by health authorities such as Food and Drug Administration (FDA) and European Medicines Agency (EMA).
- This transformation moves beyond traditional spreadsheets and basic automation to implement intelligent systems that can autonomously understand context and make decisions.
- As a result, companies are seeing significant gains in operational efficiency, improved compliance, and the elimination of data silos.
On this page
In Technology’s Grasp
In the past few years, technology has been evolving at such a fast pace that every industry has been galvanised to adopt and integrate advanced technologies into its business processes.
The end goals remain constant: resource optimisation, faster turnaround time, reduced operational cost, improved compliance and so on.
The highly regulated pharma industry is testing and implementing various technologies to streamline its processes. Adoption of technologies by regulatory health authorities such as FDA and EMEA, (e.g. Elsa, a generative artificial intelligence (AI) tool designed to help employees — from scientific reviewers to investigators — to work more efficiently with EMA specialising in developing "agentic AI" systems, which are advanced AI agents that can understand context, make decisions, and execute actions autonomously to achieve specific business goals) has accelerated the industrywide vision and confidence and transformation of the continuously explored regulatory affairs. The journey from spreadsheets to an electronic system, and from Robotic Process Automation (RPA) to AI, has led to regulatory affairs showing a high level of improvement in bringing overall efficiency (speed vs volume), eliminating data siloes, reducing human interventions (for increased productivity), and improving compliance.
Spotlighting GenAI
With the focus currently on GenAI/agentic AI, the industry is transitioning into a new era to deliver higher efficiency and innovative medicines to market earlier, with significantly reduced timelines, enabling autonomous decision-making through intelligence-embedded processes.
In the digital maturity journey, the agentic approach is being considered to replace traditional AI models, as it can think, plan, act, and take autonomous decisions. It can operate independently, collaborate across various systems, and, with the bonus of using the knowledge and experience gained.
The major difference between classical AI, GenAI, and agentic AI lies in enhanced cognitive capabilities. While AI performs rule-based execution and predefined workflows, GenAI uses knowledge to contextualise and generate text, and agentic AI uses knowledge and experience to autonomously take decisions for process orchestration and workflow execution.
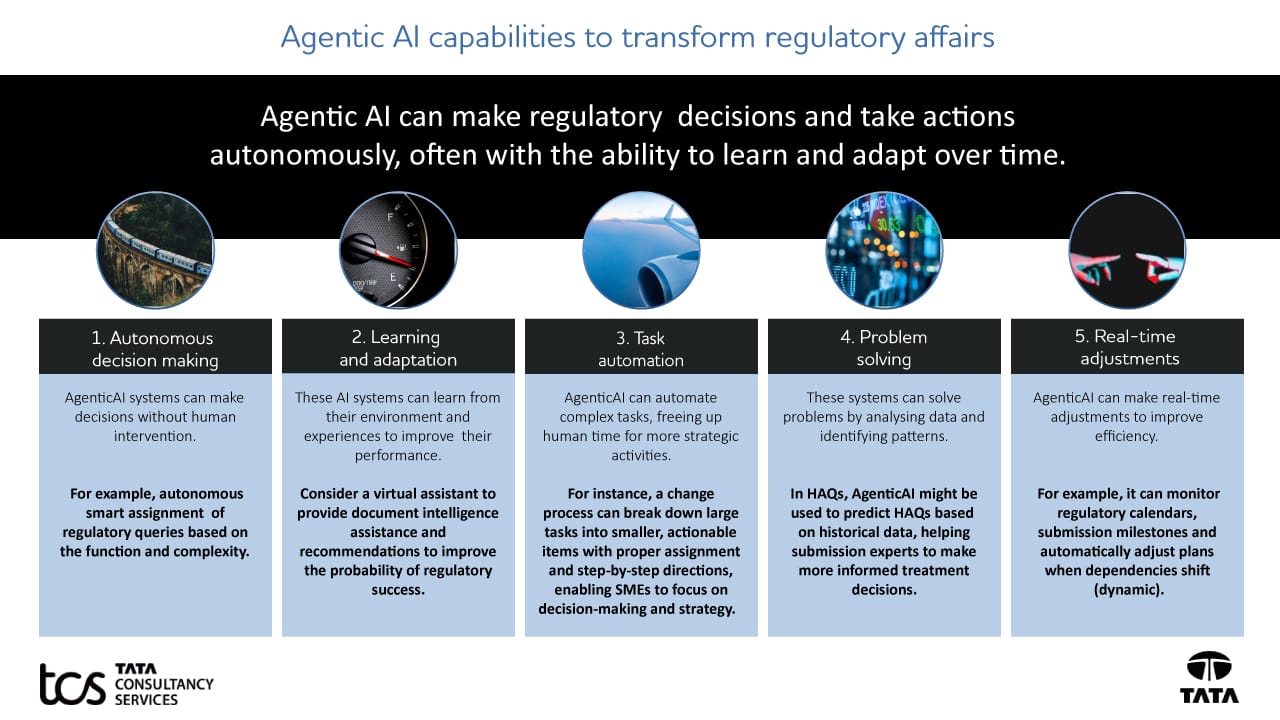
Various capabilities of agentic AI can further improve regulatory processes to deliver greater business value, as shown in the following infographic.
Divide and rule
Across the regulatory value chain, the agentic approach can further accelerate regulatory outcomes by leveraging agents with different capabilities to coordinate and orchestrate the process toward the goal.
Regulatory intelligence: With changing regulatory requirements, it is challenging to stay up to date on new guidelines and remain compliant. The agentic AI approach is to leverage multiple agents performing different tasks to enable real-time tracking of new regulatory guidelines and capture global and country-specific requirements, extract and summarise the information, assess impact, and notify relevant stakeholders and initiate workflow with task allocation to relevant stakeholders. By doing so, agentic AI can provide a real-time view of changing regulations, provide better control on compliance and competitive advantage, enabling faster turnaround time in information processing with improved resource utilisation.
Regulatory planning: Submission planning needs to be highly dynamic to improve forecasting and decision-making. Agentic AI can enable dynamic submission timelines based on historical plans, accordingly, provide allocation based on the workload, and auto-prioritise risks.
Regulatory document creation: It is a highly time-consuming process that requires significant human effort to identify sources, authors, and Quality Check (QC) content. Agentic AI can enable faster content generation and document building while identifying missing content. It can auto-classify and tag docs into the correct Electronic Common Technical Document (eCTD) module section, take content reuse decisions, autonomously pull approved text blocks and determine applicability across regions. Auto-document construction is based on the document type and relevant data. It provides low-touch, high-quality first draft document generation, requiring less effort to review.
Document assembly: Submission assembly requires high manual effort to validate and avoid technical rejections. Agents with different capabilities for checking and correcting can manage auto-compilation of eCTD sequences, check validation, handle errors, and submit to the Health Authority (HA) gateway. It autonomously fixes the common eCTD validations e.g., metadata, missing leaf, and others. It can automatically determine whether failed submissions can be resubmitted.
Health authority interactions: End-to-end health authority interaction management is complex and time-consuming, requiring monitoring, searching, and creating high-quality responses by reusing existing responses and ensuring a high success rate. Agentic AI can auto-capture/extract and assign queries based on domain, function, eCTD module, leaf, and others. It can continuously improve through feedback loops to enhance future processes.
HA commitment tracking: Determining due dates and issuing alerts to ensure compliance with HA commitments is resource-intensive. Agents can monitor, auto-classify Type 1A and 1B II variations, and independently decide whether renewal is needed based on expiry rules. It can draft responses based on queries and identify the type of file generation, e.g., the Lost Distribution Report in structured product labelling (LDR-SPL file).
Agents monitor activities, auto-classify Type 1A and 1B II variations, and assess renewal needs in accordance with expiry rules.
Safety and labelling: Compliance challenges arising from missing signals can be reduced with the agentic approach, where agents can detect them and, based on signal detection decisions, leverage human-in-the-loop to create regulatory reports.
Impact analysis on labels (Company Core Data Sheet vs local label), and propagation of CCDS updates to all local labels or only selected regions can be enabled. Periodic Safety Update Report/Periodic Benefit-Risk Evaluation Report automation, determination of inclusion/exclusion datasets based on template/Standard Operating Procedure (SOP), and live translation adaptation are a few areas where agents can help.
Master data governance: Autonomous approval/rejection of new data entries based on Identification of Medical Products (IDMP rules and historical consistency, Terminology alignment: Preferred mapping of substances, indications (media terms) and adapting auto-suggestion. Learns from authority rejections of Substance Product, Organisation and Referential (SPOR) and adapts the master data process.
Regulatory Information Management (RIM) query management: To streamline regulatory query management, agents can help with auto-capturing, classifying and responding to queries as per the complexity through appropriate channels.
Adopting agentic ai
To align with industry pace, regulatory organisations need to consider the following aspects to ensure readiness and maturity for adopting a new way of working enabled by technology.
- Trust and mindset: The trust and mindset of people in the organisation can accelerate the adoption rate. The shift can be driven by proper awareness sessions, webinars, and training.
- Infrastructure investment: In a regulatory environment, it is critical to ensure security, data protection, explainability and compliance with the outcome. The provision of guard rails in the regulatory context and governance is mandatory. Hence, proper infrastructure, which ensures data protection and security, is needed.
- Leadership buy-in: Any organisation can successfully adopt innovative approaches like agentic AI to empower their workforce, bring efficiency and ensure quality and compliance if there is a leadership stake in it. The top-down approach to integrating agentic AI like technology is highly recommended.
- Skill set orientation: Organisation-wide orientation to enhance skillsets in agentic AI can make the workforce knowledgeable, empowered, and adaptive, and can efficiently use technology in their day-to-day work. Training linked to their performance goals and rewards can help.
- Projects and experimentation: Experimentation and successful implementation of use cases can keep teams motivated and achieve confidence—for example —hackathons, ideathons, rewards and recognition.
- Knowledge management: Knowledge management is the foundation in making the agentic approach in Regulatory Affairs transformation journey successful, reduce the overall percentage of human touch to make processes highly resilient and accurate.
Agentic AI: The next leap
In the digital maturity journey, for adaptive and resilient pharma regulatory affairs, the agentic approach can be a game-changer.
As agentic AI has the capability of perceiving, reasoning, and acting autonomously to achieve objectives, making decisions without constant human input, it can help in reducing significant human effort, eliminating the burden on tacit knowledge and improving highly time-consuming processes.
The capability of agentic AI, leveraging multiple agents with specific task-oriented capabilities, collaborating in a highly coordinated environment, can enable self-optimising business processes, real-time decision-making, and the management of end-to-end workflows with minimal human oversight, while also enabling collaboration with humans for complex decision-making.
Organisations are experimenting with this technology and evaluating value generation, new investments in infrastructure building, management of security risk and governance, focus on data quality and integration, and evolving operating models, with change management being the new trend.